You are here:
- Home »
- Teaching science »
- Teaching strategies »
- Teaching with models
Teaching with models
Models are human inventions, based on an incomplete understanding of how nature works.
A model is a representation of an idea, object, event, process or system.
Models and modelling play a crucial role in science practice. One justification for their inclusion in science teaching is that they contribute to an "authentic" science education, where teaching reflects the nature of science as much as possible.
Types of models – jump to a place on this page where you can see examples for some types of models
Learning challenges when using models
Models are human inventions, based on an incomplete understanding of how nature works. Models use, as a comparison, something that is familiar to explain or describe something that is unfamiliar.
Consequently, most models are limited or "wrong" in some key aspect. Keep this is mind, as learning problems may arise if your students take a different meaning from the model than the one you intended.
For example, students may learn the model rather than the concept it is meant to illustrate. Likewise, they may lack awareness of the boundary between the model and the reality the model represents.
They may lack the necessary visual imagery to understand the model, or they may fail to distinguish between a mental image and a concrete model.
Some students may mix up aspects of two or more different models or find it difficult to apply the model in different contexts. Perhaps they miss some key attributes and thus, misunderstand the purpose of the model.
Another downfall is they may become dependent on the least sophisticated model, even when they have been introduced to more advanced models.
How to overcome learning challenges
Overcoming such learning challenges requires careful teaching intentionally focused on the model as an idea, object, event, process, or system.
A new model could be introduced in a sequence such the one that follows.
- Introduce the idea the model is intended to show. Find out what ideas students already have about that event or pattern.
- Carry out the modelling activity.
- During the activity (or at the end if more appropriate), talk about how the model/modelling activity is "like" what would really be happening and how it is "different".
- positive features of the model (what is deliberately chosen to represent "reality")
- negative features of the model (what is deliberately excluded)
- neutral features (what is ignored or not commented on).
- Return to the "big idea" at the end and let the students explain to you the sense they have made of the activity. Older students could analyse the model for themselves after some practice runs and their comparisons could be used to assess their new learning. Students may continue to need help do this for every new model used.
From mental model to expressed model
Mental models are used to describe and explain phenomena that cannot be experienced directly. Scientists use mental models to think through abstract ideas and theories.
Mental models become expressed models when they enter the public domain through action, speech, and writing. They are often represented as analogies and metaphors.
Examples of this process are:
Structure of the benzene molecule
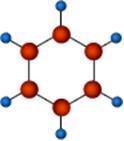
August Kekule was puzzled by benzene, a 6-carbon molecule. He pondered it for a long time.
Many stories of his famous dream emerged, specifically, the dream where he saw dancing snakes biting their own tails. From this, Kekule realised the benzene molecule could be seen as a ring structure rather than a straight chain.
This was his mental model. The expressed model he made as a result of his “dream” helped others understand how the atoms could fit together.
The composition of atoms
When John Dalton started thinking about atoms, he thought of them as if they were bowls or balls – this was his mental model.
His experiments in 1802 supported the theory that matter was made of particles, and he pictured them as small billiard balls.
Using this model, he showed how each element could be represented as being made up of the same kinds of atoms, and that compounds could be explained as being made up of atoms in specific ratios – this was his expressed model.
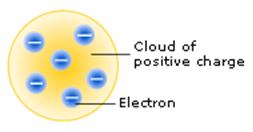 The "plum pudding" model of atom composition
The "plum pudding" model of atom composition

J.J. Thomson studied atomic theory and cathode rays and postulated the existence of small negative particles known as electrons.
He realised that the "billiard ball" model – where atoms had the same composition throughout – did not explain the existence of electrons. His expressed model showed atoms having negative electrons dotted throughout the positive atom, like plums in a pudding.
Types of expressed models
Expressed models used in science communication and teaching include:
- two-dimensional models, such as those found in textbook diagrams
- three-dimensional models such as scaled miniatures (a smaller version of large structures)
- scaled enlargements (an enlarged view of something too small to be seen)
- working models.

One example of a two-dimensional model is Leonardo da Vinci’s “Vitruvian man”.
Leonardo da Vinci created many of these wonderful, two-dimensional anatomical drawings, and in doing so, he helped further medical understanding.
Another example of a model is the internal structure of the Earth at Te Papa Tongarewa’s Awesome Forces exhibit. This model is a scaled miniature that shows the layers of the Earth’s interior in what is believed to be their correct proportions.
The structure of a cell can be represented as a scaled enlargement.
Digital models
Digital models include animated models and simulations.
Animated models may also allow students to control variables to see what impact each variable has. Simulations allow students to simulate a situation, such as making choices about land use.
Digital models intended for learning are called learning objects.
Scootle
A national repository that provides Australian schools with digital resources and where teachers can find a wide variety of learning objects suitable for use with New Zealand students of all ages.
These objects were previously available in Digistore, Te Pātaka Matahiko. They include animated models of science concepts such as cell division and tectonic plate movement, through to simulations of decisions about land use and factors which influence the UV index.

